生物医药产业是保障人类生命健康的战略性新兴产业。我国“十四五”规划明确提出构建共性基础技术供给体系,重点突破生命健康领域的关键技术瓶颈,其中生物分离技术作为获取高纯度生物活性物质的核心手段,直接关系着生物制品的质量和产业化效率[1]。
针对上述问题,本文提出通过静电纺丝技术和化学改性制备多孔氢化苯乙烯-丁二烯嵌段共聚物(SEBS)阳离子交换纤维膜。SEBS因其优异的力学性能、化学稳定性和可加工性,成为一种潜在的离子交换材料。采用溶胀-超交联处理,优化了其孔隙结构和表面化学性质,再通过磺化改性,在SEBS分子链中引入磺酸基团(—SO3H),赋予其阳离子交换能力,从而提升了蛋白质的吸附容量和传质效率。
1 实验部分
1.1 主要原料
氢化苯乙烯-丁二烯嵌段共聚物(SEBS),503T型,巴陵石油化工有限责任公司;甲苯、甲醇、三氯甲烷、1,2-二氯乙烷(DCE)、冰醋酸、三氯化铁(FeCl3)、磷酸二氢钠、磷酸氢二钠,国药集团化学试剂有限公司;去离子水,实验室自制;甲醛二甲缩醛(FDA),纯度≥99%,阿拉丁试剂(上海)有限公司;溶菌酶,生物试剂级,合肥博美生物科技有限责任公司;EX-CELL®EBxTM GRO-I无血清培养基,西格玛奥德里奇(上海)贸易有限公司;细胞计数试剂盒-8(Cell Counting Kit-8,CCK-8,XK6005S),上海晅科生物科技有限公司;SDS-PAGE电泳试剂盒(G2037-50T),塞维尔生物科技有限公司;5×SDS-PAGE蛋白上样缓冲液(G2075-1mL),塞维尔生物科技有限公司;预染蛋白Marker(G2086-205UL), 塞维尔生物科技有限公司。
1.2 SEBS纤维膜的制备
首先,将甲苯与三氯甲烷以3∶7的体积比混合制备混合溶剂,随后加入质量分数为8%的SEBS母粒,通过充分搅拌使其完全溶解,形成均一纺丝溶液。采用静电纺丝技术,在10 kV工作电压和15 cm接收距离下制备SEBS纤维膜。之后,将纤维膜浸入75%乙醇溶液中清洗,干燥后,获得洁净的SEBS纤维膜(标记为FM)。
1.3 多孔SEBS纤维膜的制备
预交联反应:将0.5 g SEBS纤维膜浸入由40 mL冰醋酸、5.6 mL甲醛二甲缩醛和10 g FeCl3组成的反应介质中,在80 ℃油浴条件下分别反应1、3、5 h。反应完成后,依次采用甲醇、1 mol/L HCl和去离子水在索氏提取器中回流清洗30 min,去除残留试剂,随后于40 ℃真空干燥24 h,得到预交联纤维膜(标记为FM-1、FM-3、FM-5)。
超交联反应:取0.5 g预交联纤维膜FM-1,在40 mL 1,2-二氯乙烷和5.6 mL甲醛二甲缩醛中溶胀12 h,随后加入10 g FeCl3,在80 ℃下分别反应1、3、5、9 h。反应结束后,经索氏提取器依次用甲醇、1 mol/L HCl和去离子水清洗,40 ℃干燥24 h,得到超交联纤维膜(标记为PFM-1、PFM-3、PFM-5、PFM-9)。
1.4 磺化多孔SEBS纤维膜的制备
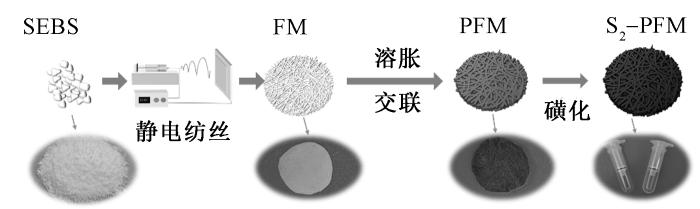
将FM、PFM-1、PFM-3、PFM-5和PFM-9纤维膜裁剪成合适尺寸,完全浸入硫酸溶液中,在98 ℃油浴条件下反应2 h,同时施加磁力搅拌以保证反应均匀性。反应完成后,取出纤维膜并用大量去离子水反复浸泡1 h,期间多次换水以彻底去除残留硫酸。随后采用乙醇清洗15 min,去除有机杂质,最后用去离子水冲洗至中性。经60 ℃烘箱干燥24 h后,得到磺化纤维膜(见图1),分别命名为S2-FM、S2-PFM-1、S2-PFM-3、S2-PFM-5和S2-PFM-9,并密封储存于干燥器中备用。
图1
图1
多孔磺化SEBS纤维膜制备流程
Fig.1
Preparation process of porous sulfonated SEBS fiber membrane
1.5 平板膜制备过程
以SEBS纤维膜纺丝溶液为铸膜液,采用50 μm厚度的刮刀刮涂,60 ℃下干燥4 h,制备得到SEBS平板膜(标记为M);采用S2-PFM-1的磺化改性工艺对该平板膜处理,获得磺化平板膜(S2-M),采用PFM-1的交联工艺和S2-PFM-1的磺化改性工艺对该平板膜处理,获得磺化多孔平板膜(S2-PM-1)。
1.6 结构及性能测试与表征
1.6.1 形貌观察
使用 JSM-65410LV 型扫描电子显微镜(SEM,日本电子JEOL公司)对SEBS纤维膜微观形貌进行观察,加速电压为 20 kV。在测试之前,对样品表面进行镀金处理 2 min。
1.6.2 比表面积测试
使用BELSORP MaxII型比表面积及孔径分析仪(日本麦奇克拜尔公司)对反应前后SEBS纤维膜比表面积进行测试。以N2为吸附质、液氮环境测试;纤维膜于120 ℃脱气4 h预处理,参照 GB/T 19587—2004《气体吸附BET法测定固态物质比表面积》,采用 BET 法计算比表面积。
1.6.3 孔隙率测试
使用AccuPycⅡ1340型全自动真密度分析仪(美国麦克仪器公司)通过气体置换法对反应前后SEBS纤维膜孔隙的率进行测试。使用氦气置换,15~35 ℃,360 s平衡,参照ASTM UOP 821—1981《使用微量分析仪通过氮气吸附和/或解吸对多孔物质进行自动微孔径分布的标准方法》进行测试。
1.6.4 化学官能团表征
使用Tensor27型傅里叶红外光谱仪(FT-IR,布鲁克(北京)科技有限公司)表征磺化前后纤维膜的化学键和官能团。测量范围为4 000~400 cm-1,分辨率为4 cm-1。
1.6.5 接触角测试
使用DSA300S型接触角测量仪器(德国克吕士公司)测量液滴在固体表面形成的接触角,评估材料的表面能及其润湿性。
1.6.6 力学性能测试
使用INSTRON 5967型拉伸测试仪(美国英斯特朗公司)测试交联前后SEBS纤维膜的拉伸性能,实时记录应力-应变曲线。观察样品的变形过程,直至样品断裂或达到测试终止条件。
1.6.7 细胞活性测试
采用 CCK-8 法检测内皮细胞活性,评估 FM、PFM-1、S2-FM、S2-PFM-1这4种纤维材料的细胞相容性。首先,在无血清培养基中对4种材料样品进行浸提,得到初始浸提液(记为100%浸提液);再对其进行不同比例的稀释,得到初始浸提液的体积分数分别为12.5%、25%、50%、100%的浸提液(记为 “材料名称-初始浸提液的体积分数”,如 FM-100%);其次,将内皮细胞接种于装填有上述浸提液的 96 孔板,在适宜条件下预培养 24 h;再向各孔中加入 CCK-8溶液,培养箱内避光培养 4 h 后获得液体产物,通过酶标仪检测各孔中液体产物在450 nm 波长处的吸光度值,通过下式计算细胞存活率细胞存活率C,以评估细胞活性。
式中:Ae为实验组液体产物的吸光度值(含细胞、材料浸提液与 CCK-8 溶液);An为阴性对照组吸光度值(含细胞、无材料浸提液的培养基与 CCK-8 溶液);Ab为空白对照组吸光度值(仅含培养基与 CCK-8 溶液)。
1.6.8 纤维膜静态吸附性能测试
本研究利用磺化纤维膜表面带负电的磺酸根与溶菌酶(pI为10~11)带正电氨基间的静电相互作用,考察纤维膜的蛋白质吸附特性。首先采用1 mol/L NaOH/NaCl溶液预处理纤维膜以活化表面基团,经去离子水充分洗涤后干燥备用。配制pH=5的磷酸盐缓冲溶液(0.2 mol/L NaH2PO4/Na2HPO4)作为模拟生理环境,并溶解溶菌酶至2 mg/mL,获得吸附前原始溶液。取50 mg预处理纤维膜浸入15 mL吸附前原始溶液,于室温下以100 r/min振荡2 h至吸附平衡。最后通过MD-2000C型紫外-可见分光光度计(北京凯奥科技发展有限公司)在280 nm处测定上清液吸光度变化,定量分析吸附性能。纤维膜对溶菌酶的吸附容量q*计算公式为
式中:C0为配制溶菌酶的初始质量浓度, g/L;C1为吸附后溶菌酶的质量浓度, g/L;V为用以吸附溶菌酶溶液的体积, L;m0为用以吸附溶菌酶的纤维膜的质量, g。
2 结果与讨论
2.1 多孔磺化SEBS纤维膜化学反应过程
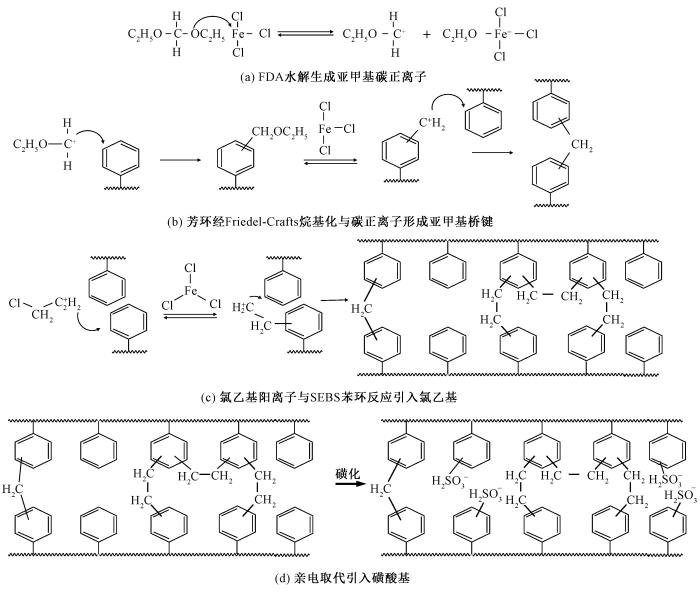
图2
图2
多孔磺化SEBS纤维膜的化学反应过程示意图
Fig.2
Schematic diagram of chemical reaction process of porous sulfonated SEBS fiber membrane. (a) Hydrolyzation of FDA to form a methylene carbocation; (b) Formation of a methylene bridge bond by aromatic ring with carbocation via Friedel-Crafts alkylation; (c) Chloroethyl cation reacts with benzene rings of SFBS to introduce chloroethyl group;(d) Introduction of sulfonic acid group via an electrophilic substitution reaction
超交联过程主要涉及以下步骤。1)反应引发。在路易斯酸催化剂(FeCl3)的作用下,1,2-二氯乙烷被活化,生成氯乙基阳离子,作为烷基化试剂。2)烷基化反应。如图 2(c)所示,氯乙基阳离子与SEBS分子链中的苯环发生Friedel-Crafts烷基化反应,在苯环上引入氯乙基(—CH2—CH2Cl)基团。由于空间位阻效应,部分DCE分子仅与1个苯环反应,而未反应的CH2Cl在后续处理中发生水解,生成羟乙基(—CH2—CH2OH)基团。甲醛二甲缩醛本身含有醚键(—OCH3),在FeCl3的催化下,可能与其它分子链中的羟基(—OH)反应,形成新的醚键(C—O—C)。3)氧化反应。在水解过程中,残留的Fe3+离子作为氧化剂,将羟乙基(—CH2—CH2OH)基团进一步氧化为羰基—CH2—C══O基团[15]。磺化过程。SEBS的磺化反应主要发生在PS嵌段的苯环上,通过亲电取代反应引入磺酸基团(—SO3H)。此过程与Friedel-Crafts烷基化交联反应形成动力学竞争,如图 2(d)所示。
2.2 预交联时间对纤维膜形貌的影响
图3
图3
SEBS纤维膜预交联不同时间的形貌照片
Fig.3
Morphology images of SEBS fiber membranes with pre-crosslinking at different time
通过对比不同预交联时间样品(FM-1、FM-3、FM-5)经溶胀-超交联1 h后形貌(见图4)发现,预交联时间延长显著提高纤维表面交联点密度,致使其形成致密皮层结构,抑制溶胀剂向纤维内部渗透,导致孔隙生成效率降低。短时预交联(如FM-1)因皮层结构疏松,利于溶胀剂扩散并形成均匀孔隙网络;而长时处理(如FM-5)因过度交联使孔隙数量锐减。基于此,将预交联时间优化至1 h(FM-1)可平衡纤维膜形貌稳定性与孔隙结构的保留,故选定FM-1作为后续研究体系。
图4
图4
FM-1、FM-3、FM-5超交联1 h的形貌照片
Fig.4
Morphology images of FM-1, FM-3,and FM-5 after hypercrosslinking for 1 h
2.3 超交联时间对纤维膜形貌的影响
图5
图5
FM-1超交联不同时间的形貌照片
Fig.5
Morphology images of FM-1 at different hypercrosslinking times
2.4 磺化对纤维膜形貌的影响
图6示出磺化SEBS纤维膜表面SEM照片。可见,磺化处理后SEBS多孔纤维膜表面形貌与原始样品相比未发生显著改变,这归因于磺化反应主要通过对分子链中苯环的化学接枝引入磺酸基团,而纤维的宏观形貌仍由静电纺丝工艺主导。交联-磺化协同处理后的纤维同样维持原有表面特征,进一步证实该过程未对物理结构造成明显影响。
图6
图6
PFM系列纤维膜磺化后的表面形貌照片
Fig.6
Surface morphology of PFM-series fibrous membranes after sulfonation
2.5 SEBS纤维膜的比表面积及孔隙率
表1示出经过不同时间的预交联与超交联后SEBS纤维膜的比表面积和孔隙率数据。实验数据显示,仅经过预交联处理的膜材料(FM系列)的比表面积(1.978 2~4.092 5 m2/g)较低。经过超交联后的膜材料(PFM系列)的比表面积(5.171 2~19.649 8 m2/g)显著提升,其中PFM-5的比表面积(19.649 8 m2/g)最高,较FM膜材料提升近10倍,同时孔隙率也同步上升至(78.76±1.27)%。然而,继续延长超交联时间至9 h,PFM-9膜材料的比表面积下降至18.663 0 m2/g,证明过高的交联密度会导致孔结构塌缩。此外,S2-PFM-1膜材料的比表面积(6.1710 m2/g)和孔隙率((65.18±2.16)%)相较于PFM-1小幅度提升,表明磺化改性并未显著改变纤维表面的孔结构,而主要改变了纤维膜表面的化学结构(见图 6)。
表1 不同试样的比表面积和孔隙率
Tab.1
| 试样编号 | 比表面积/(m2·g-1) | 孔隙率/% |
|---|---|---|
| FM | 1.987 2 | 49.27 ± 0.85 |
| FM-1 | 3.542 3 | 51.23 ± 1.54 |
| FM-3 | 3.997 8 | 51.98 ± 0.98 |
| FM-5 | 4.092 5 | 50.25 ± 1.65 |
| PFM-1 | 5.171 2 | 57.32 ± 2.72 |
| PFM-3 | 8.225 2 | 60.74 ± 1.65 |
| PFM-5 | 19.649 8 | 78.76 ± 1.27 |
| PFM-9 | 18.663 0 | 73.64 ± 1.89 |
| S2-PFM-1 | 6.171 0 | 65.18 ± 2.16 |
2.6 SEBS纤维膜的化学结构
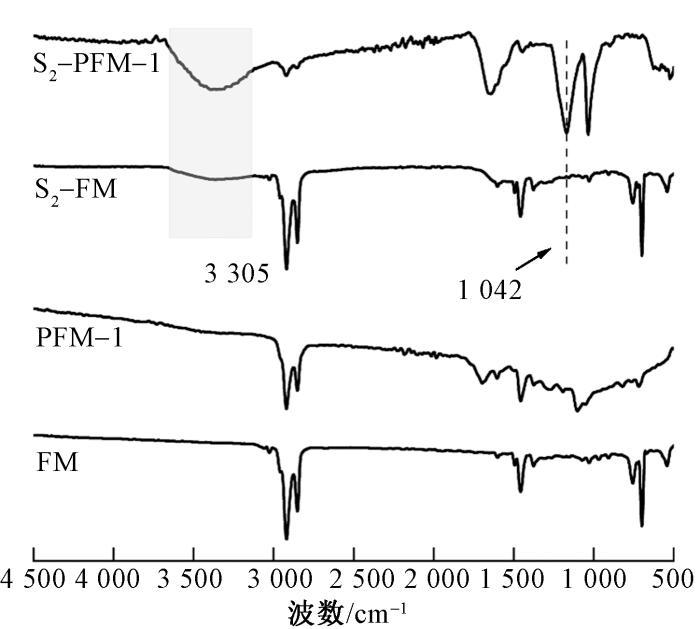
图7
图7
磺化前后纤维膜的FT-IR谱图
Fig.7
FT-IR spectra of fiber membranes before and after sulfonation
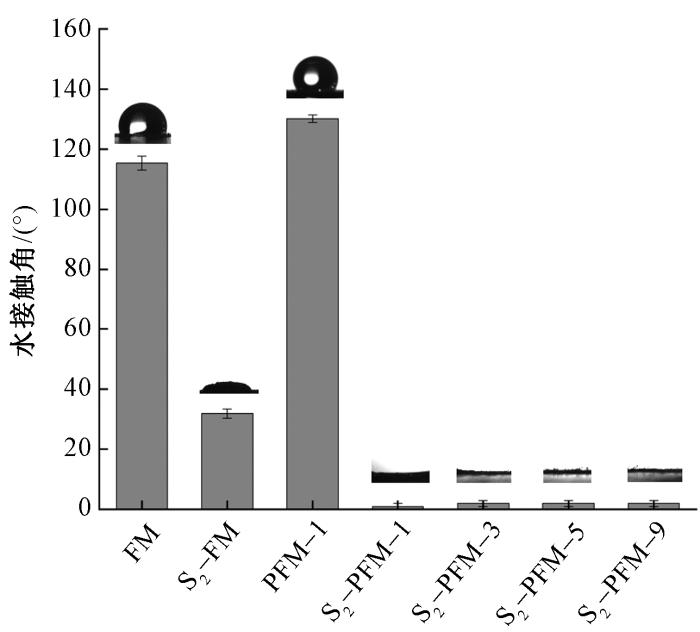
2.7 SEBS纤维膜的表面亲水性
通过接触角测试,可观察到磺化处理对SEBS纤维膜表面亲水性的显著影响(见图8)。未交联的SEBS纤维膜(S2-FM)磺化后,接触角从115°显著降低至31°,表明磺酸基团的引入大幅提高了材料的表面亲水性。然而,由于S2-FM未经过交联处理,磺化后的纤维膜在接触角测试中易被水溶胀,导致液滴形状不规则,这反映了材料在未交联状态下的结构不稳定性。相比之下,多孔SEBS纤维膜磺化后,接触角降低至接近0°。这归因于多孔结构的高比表面积和磺酸基团的协同作用,使得水能够迅速铺展并渗透到纤维膜内部。
图8
图8
磺化前后纤维膜水接触角
Fig.8
Water contact angle of fiber membranes before and after sulfonation
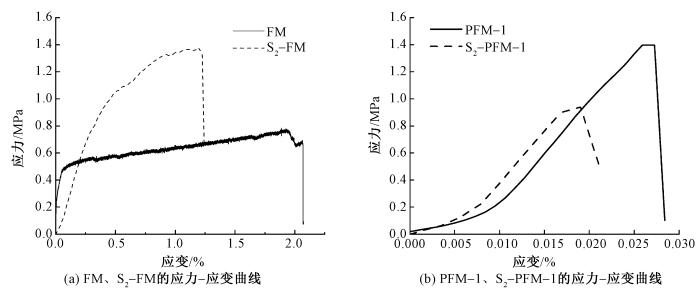
2.8 磺化多孔SEBS纤维膜的力学性能
力学性能测试表明,磺化SEBS纤维膜的拉伸应力提升源于磺酸基团引入形成的极性相互作用及氢键网络增强效应,但分子链运动受限导致材料延展性显著降低(见图9)。值得注意的是,尽管磺化后拉伸应力和应变都进一步降低,但其拉伸应力仍能达到1 MPa。其拉伸性能降低可归结于多孔基体有效承载截面积缩减与磺酸基团对分子链物理缠结的部分破坏协同作用。
图9
图9
磺化改性对 FM与 PFM-1力学性能的影响
Fig.9
Effects of sulfonation modification on mechanical properties of FM and PFM-1.(a)Stress-strain curves of FM and S2-FM; (b) Stress-strain curves of PFM-1 and S2-PFM-1
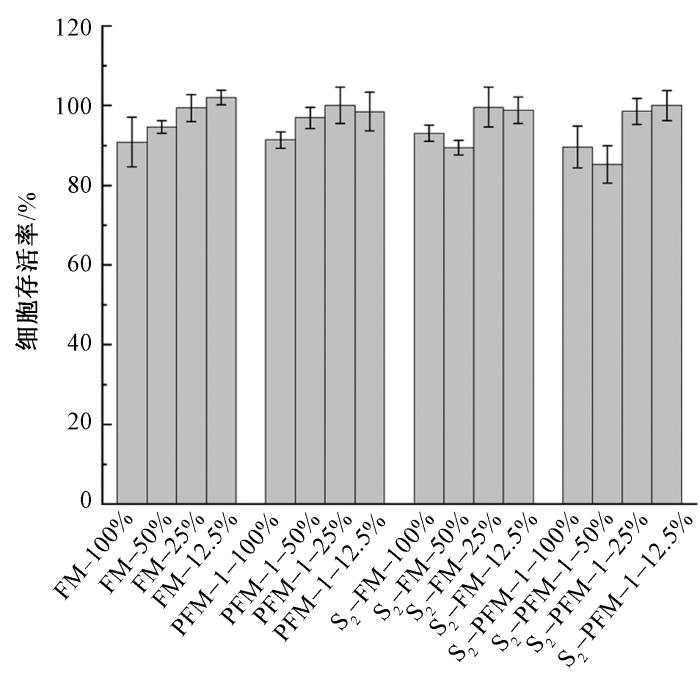
2.9 磺化多孔SEBS纤维膜的细胞相容性
经磺化后,SEBS纤维膜和多孔SEBS纤维膜的细胞存活率仍保持在80%以上(见图10),表明磺化处理未显著影响其细胞相容性。
图10
图10
不同体积分数浸取液试样的细胞存活率
Fig.10
Cell viability rate of samples with different extract concentrations
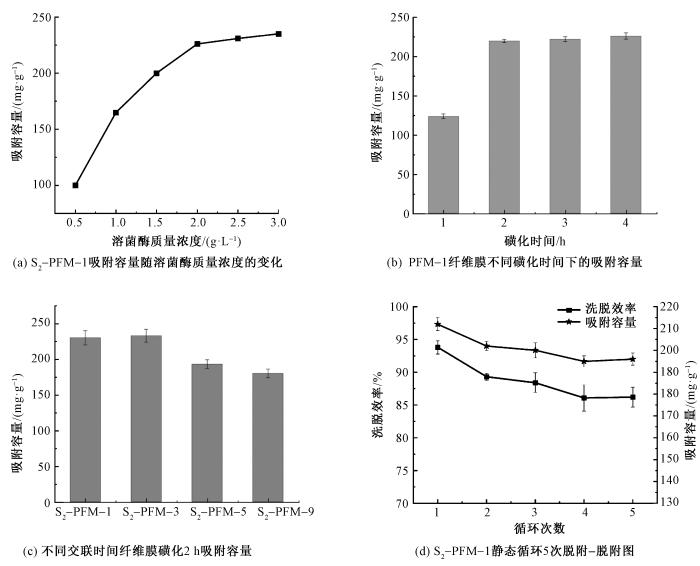
2.10 SEBS纤维膜对溶菌酶的吸附性能
为探究影响PFM系列纤维膜吸附溶菌酶能力的关键因素,实验围绕溶菌酶初始质量浓度、磺化时间、纤维膜类型及循环性能展开分析。在溶菌酶初始质量浓度影响方面,随浓度升高,纤维膜吸附容量显著提升,当质量浓度达 2 g/L 时,吸附容量达 226 mg/g;继续增加质量浓度,吸附容量无明显变化,表明纤维膜吸附位点接近饱和(见图11(a)),故后续实验选用 2 g/L 溶菌酶质量浓度。磺化时间影响实验结果(见图11(b))显示,超 2 h 后的吸附容量不再显著增加,因苯环上磺酸根接枝位点已完全接枝,因此确定 2 h 为最佳磺化时间。对比 S2-PFM-1 与 S2-PFM-3的吸附性能(见图11(c)),后者吸附容量虽略有提升,但差异不显著,故选择S2-PFM-1 进行后续研究。此外,交联时间过长会导致吸附容量降低,原因是过度交联占用苯环活性位点,减少磺酸根基团接枝密度(见图2)。循环性能测试(见图 11(d))中,S2-PFM-1 纤维膜经 5 次吸附-脱附循环后,吸附容量仍保持初始值的 86.2%,具备良好稳定性,满足实际应用需求。
图11
图11
SEBS纤维膜对溶菌酶吸附容量影响因素及静态循环性能
Fig.11
Factors influencing adsorption capacity of SEBS-based fiber membrane materials for lysozyme and their static cyclic adsorption-desorption performance. (a) Variation of adsorption capacity of S2-PFM-1 with lysozyme concentration; (b) Adsorption capacity of PFM-1 fiber membranes at different sulfonation time; (c) Adsorption capacity of fiber membranes with different crosslinking time after 2-hour sulfonation; (d) Static adsorption-desorption diagram of S2-PFM-1 over 5 cycles
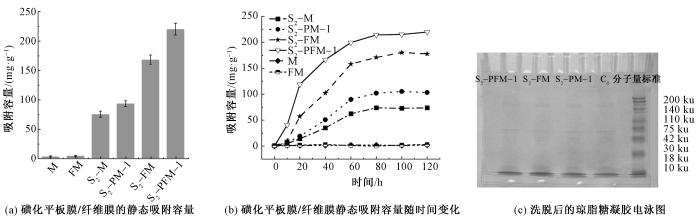
2.11 不同膜材料溶菌酶吸附性能对比
由平板膜SEM照片可以看出,平板膜交联后和磺化后的孔隙结构并不明显(见图12)。通过对比可发现,磺化多孔纤维膜对溶菌酶的吸附量显著高于磺化平板膜。
图12
图12
M、FM、S2-M、S2-PM-1、S2-FM、S2-PFM-1表面形貌对比图
Fig.12
Comparison of surface morphologies of M,FM, S2-M, S2-PM-1, S2-FM, and S2-PFM-1
磺化多孔纤维膜的高吸附量主要源于2方面:
图13
图13
平板膜与纤维膜材料溶菌酶吸附性能对比
Fig.13
Comparison of lysozyme adsorption performance between flat membrane and fiber membrane materials. (a) Static adsorption capacity of sulfonated flat film/fiber film; (b) Static adsorption capacity of sulfonated flat film/fiber film over time; (c) Eluted agarose gel electrophoresis electropherogram
3 结论
本研究制备了氢化苯乙烯-丁二烯嵌段共聚物(SEBS)纤维膜材料,并基于Friedel-Crafts烷基化反应和磺化反应,成功制备了磺化多孔SEBS阳离子交换纤维膜(S2-PFM-1)。形貌及孔结构分析结果显示,磺化后的膜材料仍保持6.171 0 m2/g的比表面积及(65.18 ± 2.16)%的孔隙率,表明磺酸基团通过分子链修饰实现接枝而未破坏三维网络结构。红外光谱分析结果显示,膜材料表面成功接枝了磺酸基团,水接触角由未磺化样品的118°显著降低至0°左右,表现出优异的亲水性能。同时,磺化后膜材料表现出较高的细胞存活率,为(89.61±5.25)%。膜材料在pH=5条件下120 min对溶菌酶的吸附达到饱和容量226 mg/g,且5次吸附-脱附循环后容量保持率仍达86.2%。交联时间通过影响苯环活性位点分布及磺酸基接枝密度,成为调控吸附性能的关键参数。综上所述,磺化多孔SEBS纤维膜表现出优异的亲水性和蛋白质吸附性能,可用于蛋白质的高效分离,在生物医药领域具有潜在的应用前景。
参考文献
High-performance separation of protein by the affinity adsorbent of polyethyleneimine-functionalized collagen fibers
[J].DOI:10.1016/j.seppur.2025.131754 URL [本文引用: 1]
Challenges and advances in the fabrication of monolithic bioseparation materials and their applications in proteomics research
[J].
Emerging nanoporous materials for biomolecule separation
[J].DOI:10.1002/adfm.v32.20 URL [本文引用: 1]
Ultrafast molecular separation through all-biomaterial membranes with a hierarchical configuration and mechanism study
[J].DOI:10.1016/j.seppur.2024.131338 URL [本文引用: 1]
Isolation of exosomes from whole blood by integrating acoustics and microfluidics
[J].
DOI:10.1073/pnas.1709210114
PMID:28923936
[本文引用: 1]

Exosomes are nanoscale extracellular vesicles that play an important role in many biological processes, including intercellular communications, antigen presentation, and the transport of proteins, RNA, and other molecules. Recently there has been significant interest in exosome-related fundamental research, seeking new exosome-based biomarkers for health monitoring and disease diagnoses. Here, we report a separation method based on acoustofluidics (i.e., the integration of acoustics and microfluidics) to isolate exosomes directly from whole blood in a label-free and contact-free manner. This acoustofluidic platform consists of two modules: a microscale cell-removal module that first removes larger blood components, followed by extracellular vesicle subgroup separation in the exosome-isolation module. In the cell-removal module, we demonstrate the isolation of 110-nm particles from a mixture of micro- and nanosized particles with a yield greater than 99%. In the exosome-isolation module, we isolate exosomes from an extracellular vesicle mixture with a purity of 98.4%. Integrating the two acoustofluidic modules onto a single chip, we isolated exosomes from whole blood with a blood cell removal rate of over 99.999%. With its ability to perform rapid, biocompatible, label-free, contact-free, and continuous-flow exosome isolation, the integrated acoustofluidic device offers a unique approach to investigate the role of exosomes in the onset and progression of human diseases with potential applications in health monitoring, medical diagnosis, targeted drug delivery, and personalized medicine.
Development of composite electrolyte membranes with functional polymer nanofiber frame-works
[J].
DOI:10.1038/s41428-024-01007-3
[本文引用: 1]

Solid electrolyte membranes based on polymers have shown promise owing to their high-energy demand and the sustainable and cost-effective nature of these materials. However, polymer electrolyte membranes composed of a polymer matrix have not progressed for the following reasons: (1) the low ion conductivity of polymer materials cannot achieve the level required for practical use, and (2) it is difficult to satisfy both battery performance and membrane durability simultaneously because of the trade-offs between ion conductivity and membrane stability. In recent years, research on composite electrolyte membranes composed of polymer nanofibers and a polymer matrix has attracted significant interest because of their improved ion conductivity, excellent membrane durability, and ability to fabricate thinner membranes. Polymeric nanofiber-containing polymer electrolyte membranes are expected to be applied not only to electrolyte membranes for fuel cells and water electrolysis, including alkaline-type electrolyte membranes for water electrolysis, but also to all-solid-state Li-ion batteries and all-solid-state Li-air batteries. This focus review presents the latest information on these topics.
Quaternized nanofiber-based anion-exchange chromatography membrane with periodic diagonal surface structure for efficient protein separation
[J].
Solution viscosity-mediated structural control of nanofibrous sponge for RNA separation and purification
[J].DOI:10.1002/adfm.v32.20 URL [本文引用: 1]
Thermally and chemically stable isoporous block copolymer membranes
[J].
DOI:10.1021/acsmacrolett.2c00352
PMID:36048420
[本文引用: 1]

Ultrafiltration (UF) membranes, particularly membranes fabricated from self-assembled diblock copolymers, hold promise in wastewater treatment, dairy, and food industries. Membrane development goals involve combining a highly porous selective layer with a narrow pore size distribution with a mechanically stable supporting layer to achieve constant flux. To date, isoporous integral asymmetric membranes have been formed either as flat sheets or hollow fibers, and a surface-selective layer determines membrane separation performance. A unique isoporous membrane of the poly(4-vinylbenzocyclobutene)--poly(4-vinylpyridine) (PVBCB--P4VP) diblock copolymer with a substructure of almost homogeneous porosity throughout the body of the material (three-dimensional porosity) has been developed. Moreover, the matrix of the membrane (PVCB) enables it to undergo cross-linking, allowing the membrane to be thermally sterilized and applied in high-temperature UF applications.
State of the art on the separation and purification of proteins by magnetic nanoparticles
[J].
DOI:10.1186/s12951-023-02123-7
[本文引用: 1]

The need for excellent, affordable, rapid, reusable and biocompatible protein purification techniques is justified based on the roles of proteins as key biomacromolecules. Magnetic nanomaterials nowadays have become the subject of discussion in proteomics, drug delivery, and gene sensing due to their various abilities including rapid separation, superparamagnetism, and biocompatibility. These nanomaterials also referred to as magnetic nanoparticles (MNPs) serve as excellent options for traditional protein separation and analytical methods because they have a larger surface area per volume. From ionic metals to carbon-based materials, MNPs are easily functionalized by modifying their surface to precisely recognize and bind proteins. This review excavates state-of-the-art MNPs and their functionalizing agents, as efficient protein separation and purification techniques, including ionic metals, polymers, biomolecules, antibodies, and graphene. The MNPs could be reused and efficaciously manipulated with these nanomaterials leading to highly improved efficiency, adsorption, desorption, and purity rate. We also discuss the binding and selectivity parameters of the MNPs, as well as their future outlook. It is concluded that parameters like charge, size, core–shell, lipophilicity, lipophobicity, and surface energy of the MNPs are crucial when considering protein selectivity, chelation, separation, and purity.
Polyethyleneimine assisted-two-step polymerization to develop surface imprinted cryogels for lysozyme purification
[J].DOI:10.1016/j.colsurfb.2016.06.060 URL [本文引用: 1]
Purification and characterization of recombinant human lysozyme from eggs of transgenic chickens
[J].DOI:10.1371/journal.pone.0146032 URL [本文引用: 1]
Friedel-Crafts烷基化反应研究进展
[J].
Progress in research on Friedel-Crafts alkylation reac-tion
[J].
Stepwise crosslinking: a facile yet versatile conceptual strategy to nanomorphology-persistent porous organic polymers
[J].DOI:10.1002/adma.v29.27 URL [本文引用: 2]
Polymeric molecular sieve membranes via in situ cross-linking of non-porous polymer membrane templates
[J].DOI:10.1038/ncomms4705 [本文引用: 1]
PP含量及纺丝条件对SEBS/PP共混料性能及可纺性的影响
[J].
Effect of PP content and spinning conditions on the performance and spinnability of SEBS/PP blends
[J].
Tailoring nanoporous-engineered sponge fiber molecular sieves with ternary-nested architecture for precise molecular separation
[J].
DOI:10.1021/acsnano.1c04575
PMID:34339181
[本文引用: 1]

Polymeric fiber molecular sieves (PFMs) with ultrahigh surface areas, well-defined Murray's-law hierarchical nanoporous structures, and superior self-standing properties are of great interest for molecular-level separation applications. However, creating such PFMs has been proven extremely challenging. Herein, we report a cross-scale pore-forming strategy to create intriguing sponge fiber molecular sieves with hierarchical, tailorable, and molecularly defined nanoporosity by nanospace-confined chain-packing modulation at the molecular level. Robust secondary ultramicropores (<7 Å) and micropores (<2 nm) are constructed in the macro/mesoporous skeletons of sponge fibers to realize a tunable pore size distribution. The resultant PFMs exhibit the integrated properties of ultrahigh surface area (860 m g), large pore volume (0.6 cm g), self-standing properties, and excellent molecular sieving performance and are widely applied in acetophenone/phenyl ethanol separation, hydrogen peroxide purification, ethyl acetate separation, and CO adsorption fields. The fabrication of such PFMs provides a feasible way for the design and development of polymeric fibrous sieves for molecular separation in large-scale chemical, energy, and environmental operation processes.
Removal of metal ions from aqueous solutions using carboxymethyl cellulose/sodium styrene sulfonate gels prepared by radiation grafting
[J].
DOI:S0144-8617(16)31103-1
PMID:27987936

Sodium Carboxymethyl Cellulose (CMCNa)/Sodium Styrene Sulfonate (SSS) hydrogels with grafted and crosslinked polymeric networks were prepared by γ-radiation at atmosphere condition. The obtained hydrogels were characterized by gel fraction, swelling ratio, TGA and FTIR spectroscopy. The results showed the ratio of CMC and SSS 1:0 gave the highest gel fraction, compared with other ratios. The swelling capacity increased by increasing SSS content due to the presence of SONa, OH groups in gel structure. The FTIR spectrum of CMC/SSS gel showed the new absorption peaks at 1034 and 1012cm corresponds to SONa group. The metal ion adsorption capacity of CMC/SSS gel was investigated. The grafted gel effectively removed metal ions, especially Cr and Pb. The effects of hydrogel composition, contact time, and initial concentration on the adsorption capacity of the grafted hydrogels were studied. The adsorption kinetics and equilibrium isotherms were investigated using pseudo-second-order model and Langmuir model.Copyright © 2016. Published by Elsevier Ltd.










 京公网安备11010502044800号
京公网安备11010502044800号